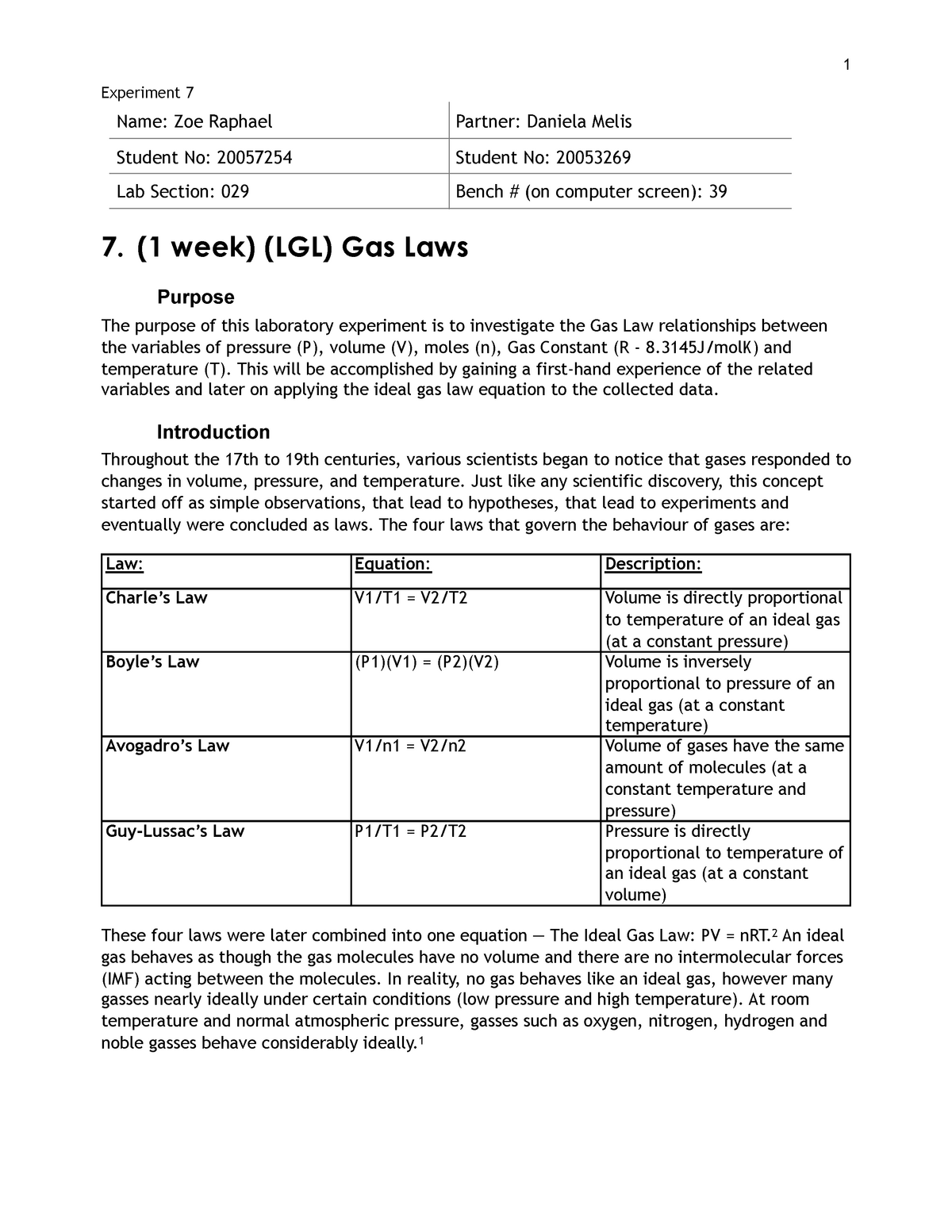
Boyle S Law Experiment Report

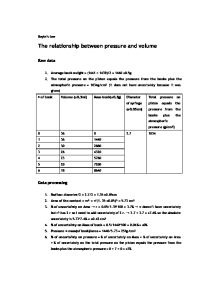
This essentially means that the pressure multiplied by the volume is constant.
Boyle s law experiment report. From the graphs it can be concluded that the pressure of the gas in the column is inversely proportional to the volume of the gas at a constant temperature for a fixed amount of gas. The experiment is very simple. We d love to have your feedback. It must likewise be total and informative.
Boyle s law states that for constant temperature the product of the volume and the pressure of an ideal gas is a constant. P 2 is the final pressure exerted by the gas. José popoff ana valeria hernández stephanye cruz introduction boylw s law is what describes the relationship between the pressure and the volume of a gas when its temperature. Where p 1 is the initial pressure exerted by the gas.
Boyle s law lab report laboratory reports are utilized to explain the study results. This law can be expressed mathematically as follows. Boyle s law was derived from a scientist named robert boyle who found that at a constant temperature the pressure of a given gas was inversely proportional to its volume. They must be readable and need to be provided in a clear and succinct manner.
The experiment is successfully studied and the system obeys boyle s law. Lauren odom morgan simms purpose. P 1 v 1 p 2 v 2. Boyle s law lab report by stephanye cruz sunshine christian bilingual institute lab report boyle s law presented for our physics class mr.
Pv c 1 the ideal gas law pv nrt 2 states that this constant nrt is proportional to the amount of ideal gas in the sam ple the number of moles n and the absolute temperature t. Boyle s law lab manual. Report an information security concern. V 2 is the final volume occupied by the gas.
Lab report boyles law yang latestttttt dut. The law was discovered by robert boyle in the 17th century. The law can be empirically proven. It states the pressure of a fixed amount of a gas is inversely proportional to its volume at a constant temperature.
This expression can be obtained from the pressure volume relationship suggested by boyle s law. The article discusses an experimental method to verify the law using a syringe. Boyle s law experiment 1. Boyle s law experiment date.